 For example, a sulfur atom (3 s 23 p 4) has a covalent radius of 104 pm, whereas the ionic radius of the sulfide anion (3 s 23 p 6) is 170 pm. Both effects (the increased number of electrons and the decreased Z eff) cause the radius of an anion to be larger than that of the parent atom ( ). This results in a greater repulsion among the electrons and a decrease in Z eff per electron. Proceeding down the groups of the periodic table, we find that cations of successive elements with the same charge generally have larger radii, corresponding to an increase in the principal quantum number, n.Īn anion (negative ion) is formed by the addition of one or more electrons to the valence shell of an atom. The radius for a cation is smaller than the parent atom (Al), due to the lost electrons the radius for an anion is larger than the parent (S), due to the gained electrons.Ĭations with larger charges are smaller than cations with smaller charges (e.g., V 2+ has an ionic radius of 79 pm, while that of V 3+ is 64 pm). The transition elements, on the other hand, lose the ns electrons before they begin to lose the ( n – 1) d electrons, even though the ns electrons are added first, according to the Aufbau principle. As a general rule, when the representative elements form cations, they do so by the loss of the ns or np electrons that were added last in the Aufbau process. Thus, as we would expect, the outermost or valence electrons are easiest to remove because they have the highest energies, are shielded more, and are farthest from the nucleus. The stronger pull (higher effective nuclear charge) experienced by electrons on the right side of the periodic table draws them closer to the nucleus, making the covalent radii smaller. Thus, Z eff increases as we move from left to right across a period. Thus, each time we move from one element to the next across a period, Z increases by one, but the shielding increases only slightly. Core electrons are adept at shielding, while electrons in the same valence shell do not block the nuclear attraction experienced by each other as efficiently. Shielding is determined by the probability of another electron being between the electron of interest and the nucleus, as well as by the electron–electron repulsions the electron of interest encounters. Covalent Radii of the Halogen Group Elements The trends for the entire periodic table can be seen in. This trend is illustrated for the covalent radii of the halogens in and. Consequently, the size of the atom (and its covalent radius) must increase as we increase the distance of the outermost electrons from the nucleus. Thus, the electrons are being added to a region of space that is increasingly distant from the nucleus. We know that as we scan down a group, the principal quantum number, n, increases by one for each element. We will use the covalent radius ( ), which is defined as one-half the distance between the nuclei of two identical atoms when they are joined by a covalent bond (this measurement is possible because atoms within molecules still retain much of their atomic identity). However, there are several practical ways to define the radius of atoms and, thus, to determine their relative sizes that give roughly similar values. The quantum mechanical picture makes it difficult to establish a definite size of an atom. They are (1) size (radius) of atoms and ions, (2) ionization energies, and (3) electron affinities. These properties vary periodically as the electronic structure of the elements changes. 
An understanding of the electronic structure of the elements allows us to examine some of the properties that govern their chemical behavior. 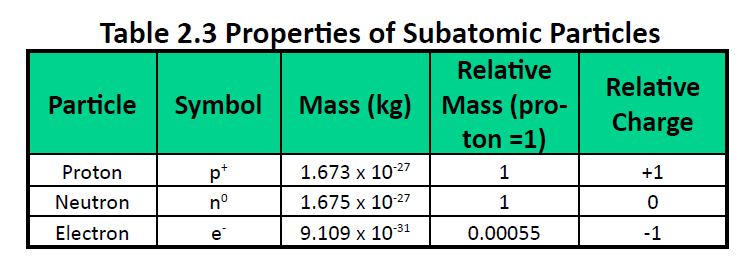
As we go down the elements in a group, the number of electrons in the valence shell remains constant, but the principal quantum number increases by one each time. Oxygen, at the top of group 16 (6A), is a colorless gas in the middle of the group, selenium is a semiconducting solid and, toward the bottom, polonium is a silver-grey solid that conducts electricity.Īs we go across a period from left to right, we add a proton to the nucleus and an electron to the valence shell with each successive element. For example, as we move down a group, the metallic character of the atoms increases. However, there are also other patterns in chemical properties on the periodic table. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. Describe and explain the observed trends in atomic size, ionization energy, and electron affinity of the elements.\): Periodic Table of Elements that is color coded for atomic mass.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
